With the increasing demand for sustainable energy solutions, all-solid-state potassium metal batteries (PMBs) are gaining significant interest as a viable alternative to conventional lithium-based systems owing to the natural abundance and high theoretical energy density of potassium. Nevertheless, the advancement of efficient PMBs is still hindered by the lack of suitable electrolytes.
Recently, in a study published in Journal of the American Chemical Society, a research group led by Prof. CHEN Ping, Prof. HE Teng, and Assoc. Prof. YU Yang, from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS), in collaboration with Assoc. Prof. WU Anan from Xiamen University, has developed a series of alkali metal pyridonate salts. Among these, the meta-substituted potassium pyridonate (meta-KOC5H4N) exhibits a high potassium ion conduction and serve as a potassium-ion solid-state electrolyte.
The research group has been dedicated to the study of metalorganic compounds for hydrogen storage. As research progressed, the group discovered that certain metalorganic compounds exhibit two-dimensional layered or three-dimensional framework structures formed by anions, which facilitate cation transport channels. However, strong cation-anion interactions or the lack of cation vacancies often impedes efficient ion conduction.
In this work, the researchers employed a molecular design approach by introducing nitrogen atoms into the phenol ring to create an unsaturated coordination environment for potassium ions. By tuning the substitution position (ortho, meta, para), three isomeric potassium pyridonates were synthesized (as shown in the figure). Among these, meta-KOC5H4N containing 1% potassium vacancies achieved an ionic conductivity of 0.22 mS cm-1 at 90°C and an ionic transference number as high as 0.99, without the need for neutral ligand assistance. Computational results indicate that the efficient ion conduction is achieved via a synergistic effect: (i) diffusion through weakly coordinated anion channels and (ii) defect-enabled interstitial hopping synergistic mechanism.
Beyond conductivity, this material exhibits exceptional thermal stability (>240°C), air tolerance, non-flammability, and a low Young’s modulus (5.84 GPa), enabling direct scalable synthesis, ease of moulding, and superior interfacial stability. The compound can be readily cold-pressed into desired forms, demonstrating excellent processability and potential for large-scale production. Furthermore, an all-solid-state PMB prototype using meta-KOC5H4N was also effectively assembled.
“This work opens up a new avenue for the design of safe and readily processable organic electrolyte materials.” said Prof. HE.
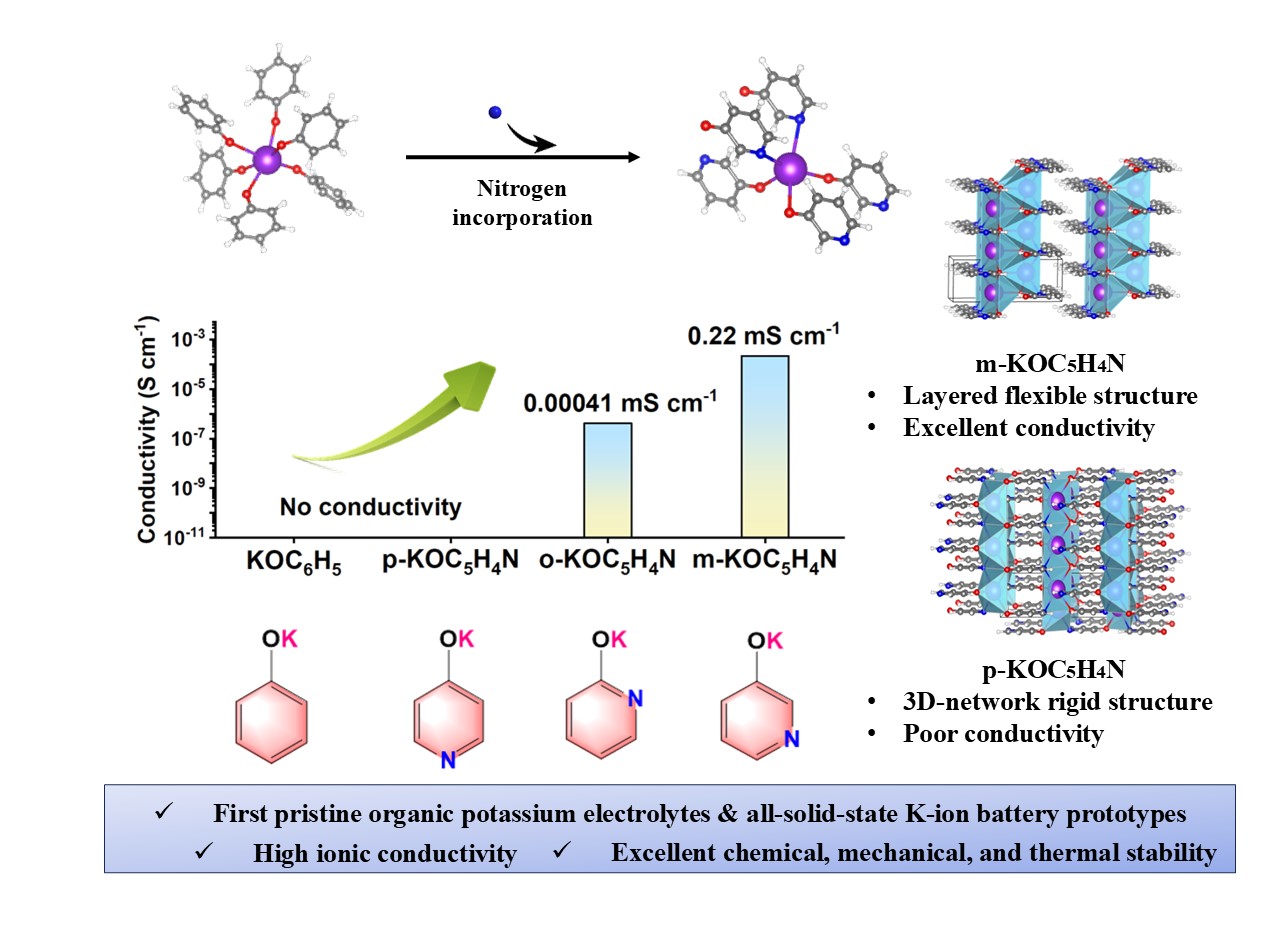
Potassium pyridonates as SSEs in potassium metal batteries.
Article link: https://doi.org/10.1021/jacs.5c19440
